Using words to describe the relationship between variables in gay lussac's law:
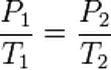
The formula for Gay-Lussac's gas law is P1/T1=P2/T2. Where P stands for pressure and T stands for temperature!
The equation shows that the initial pressure is divided by the initial temperature (in kelvins), which then equals a resulting pressure divided by the resulting temperature. The directly proportional pressures and volumes will equal the same value before and after a variable is changed. The constant in Gay-Lussac's law is the volume of the container.
The equation shows that the initial pressure is divided by the initial temperature (in kelvins), which then equals a resulting pressure divided by the resulting temperature. The directly proportional pressures and volumes will equal the same value before and after a variable is changed. The constant in Gay-Lussac's law is the volume of the container.
How did gay-Lussac use balloons to study chemistry inthe early 1800's? What did he learn from investigations he carried out using hot air balloons?

In 1804, Joseph Gay-Lussac made a hot-air balloon ascent with Jean-Baptiste Biot. They reached a height of 6.4 kilometers so that they could study the earth's atmosphere. They wanted to collect samples of air from different altitudes for analysis of the differences in temperature, pressure, and humidity. The next year he told of his findings that the earth's atmosphere does not change in composition with increasing altitude (and therefore decreasing pressure). In 1808 Gay-Lussac announced another big discovery: Gay-Lussac’s Law. Today we use Gay-Lussac's law in determining pressure and temperature differences of gas samples.
In 1804 Gay-Lussac made several other ascents of over 7,000 meters above sea level in hydrogen-filled balloons. This allowed him to investigate other aspects of gases. He not only gathered magnetic measurements at various altitudes, but he also took pressure, temperature, and humidity measurements and samples of air, which he later analyzed chemically.
In 1805, together with his friend and scientific collaborator Alexander von Humboldt, he discovered that the basic composition of the atmosphere does not change with decreasing pressure (increasing altitude).
In 1804 Gay-Lussac made several other ascents of over 7,000 meters above sea level in hydrogen-filled balloons. This allowed him to investigate other aspects of gases. He not only gathered magnetic measurements at various altitudes, but he also took pressure, temperature, and humidity measurements and samples of air, which he later analyzed chemically.
In 1805, together with his friend and scientific collaborator Alexander von Humboldt, he discovered that the basic composition of the atmosphere does not change with decreasing pressure (increasing altitude).
Sources:
✿ http://chemed.chem.wisc.edu/chempaths/GenChem-Textbook/Gay-Lussac-s-Law-952.html
✿ http://www.chemteam.info/GasLaw/Gas-Gay-Lussac.html
✿ http://answers.yahoo.com/question/index?qid=1006060205516
✿ http://www.chemheritage.org/discover/online-resources/chemistry-in-history/themes/early-chemistry-and-gases/gay-lussac.aspx
✿ http://www.chemistryexplained.com/Fe-Ge/Gay-Lussac-Joseph-Louis.html
✿ http://upload.wikimedia.org/math/8/4/9/849135802b297470cf60ce8e8d9297a6.png
✿ http://www.chemheritage.org/Discover/Online-Resources/Chemistry-in-History/Themes/Early-Chemistry-and-Gases/asset_upload_file954_36854_thumbnail.jpg
✿ http://honorsph.startlogic.com/physciseries/pss/images/gayluc.gif
✿ http://www.daviddarling.info/images/Gay-Lussac.jpg
✿http://www.indianapolisballoonrides.com/images/carousel/indianapolis-balloon-rides-01.jpg
